Call for Proposals
Home Ultrasound Implementation Pilot
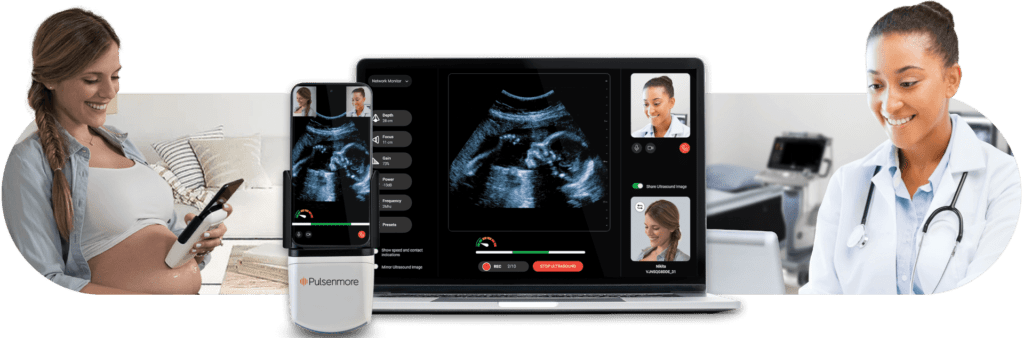
Background: Pulsenmore pioneers cutting-edge home ultrasound technology, enabling pregnant mothers to conduct self-scans with remote transmission of imaging clips to clinicians for fetal well-check diagnosis.
To familiarize yourself with the Pulsenmore solution, please visit this page for full background information.
This call for proposals aims to facilitate clinical research in prenatal care using the Pulsenmore ultrasound solution. We are interested in supporting pilot studies that investigate the potential implementation of our technology to maximize benefits and impact for patients, healthcare providers, and healthcare systems.
We invite clinicians across Europe to express their interest in receiving support from Pulsenmore by filling out the form below by May 30th 2024. Submitted forms will be reviewed by the Pulsenmore clinical team, and selected applicants will be contacted to submit the full proposal by June 27th 2024. Pilot kick-off will be conducted in the second half of 2024, with successful candidates being supported with devices, training, and pilot infrastructure.
For more information, please reach out to Dr. Ronit Shtrichman by email: [email protected]

How can I get Pulsenmore?
Pulsenmore is provided through participating healthcare providers. Ask your doctor if Pulsenmore is part of their prenatal care offering. If they work with us, they can prescribe the device, allowing you to purchase it and start using the service as part of your pregnancy care.
Is it for any pregnancy?
Pulsenmore is designed for use from week 14 of pregnancy and for single-fetus pregnancies. To ensure accurate monitoring and continuous clinical feedback, each device is personal and intended to be used only by you throughout your current pregnancy, keeping you securely connected to your care team.
Why should I buy it?
Because it gives you peace of mind when you need it most. Pulsenmore lets you check on your baby from the comfort of your home, while staying connected to real medical care. It’s the only at-home ultrasound solution where every scan is reviewed by qualified medical professionals, so you get trusted clinical insight, not uncertainty.
Is the technology safe?
Yes. Pulsenmore is based on the same ultrasound technology used routinely in clinics. Scans are performed within established safety guidelines and reviewed by a medical professional, so you receive reliable clinical insight while maintaining the safety of both you and your baby.
Can I scan without medical interpretation?
Every scan requires medical interpretation, and its important to wait until the files are fully uploaded. You are not expected to understand or interpret the ultrasound scans; follow the app’s instructions to scan, and receive a response from skilled ultrasound professionals .
Is the device reliable? How can I scan myself if I’m not a professional sonographer or obstetrician?
Yes, the device is reliable and was tested, validated and approved for use in the US, Europe, Australia, Brazil, Switzerland, the UK, Colombia and Israel. Over 200,000 successful scans have been performed since the launch of the technology. These numbers demonstrate that women can confidently scan themselves, as the diagnoisis it self is being done by a trained clinical team. The remote clinical team can accurately identify parameters related to fetal vitality, such as fetal heartbeat, fetal movements, and amniotic fluid volume, based on these scans.
How do I start working with Pulsenmore?
To begin working with Pulsenmore, clinicians or healthcare organizations can submit a request through the contact form and indicate their interest in a collaboration. Our team will follow up to introduce the system, review clinical and operational needs, and confirm alignment with your care model.
Once aligned, a service agreement is established. From that point, eligible patients can obtain the device through authorized channels and receive remote monitoring services as part of the clinician’s care offering.
Does this replace scheduled ultrasounds?
No. Pulsenmore does not replace diagnostic or scheduled in-clinic ultrasound examinations.
How does this integrate into the medical record?
Pulsenmore supports secure access to scan data, which can be referenced or documented in the EMR in accordance with institutional policies.
How do remote scans integrate into risk-based prenatal care?
Pulsenmore is used as an adjunct within physician-defined care pathways. Use is determined by patient risk profile and clinical judgment and does not replace standard prenatal imaging schedules.
What data does Pulsenmore collect?
Pulsenmore collects four types of data:
• Service logs: Application usage logs, such as performance logs and errors.
• Technical information about the smartphone: Smartphone capabilities, application version, language, etc.
• Clinical information: Ultrasound clips scan time (see Privacy Statement)
• Interpreter Data: The identifiers of the clinicians that interpret the scans.me, scan measurements and last menstrual period (LMP).
Is it safe to use Pulsenmore?
The Pulsenmore was designed to follow ALARA (As Low As Reasonably Achievable) principles as follows:
The system supports only low frequency (2-5MHz), the duration of the scan is limited up to 3 minutes in the App guided mode, or at the clinician’s discretion in the clinician guided mode, only clinician can modify the ultrasound parameters (i.e., depth, gain, frequency etc.), each scan is enabled only upon the approval of a medical professional. The system was authorized by FDA through the de novo classification process.
