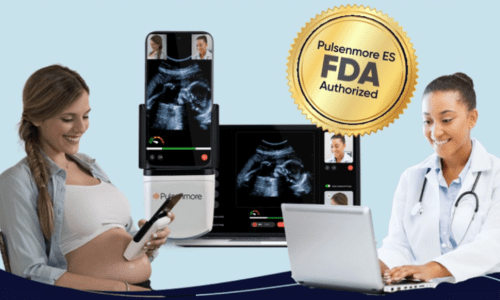
To begin working with Pulsenmore, clinicians or healthcare organizations can submit a request through the contact form and indicate their interest in a collaboration. Our team will follow up to introduce the system, review clinical and operational needs, and confirm alignment with your care model.
Once aligned, a service agreement is established. From that point, eligible patients can obtain the device through authorized channels and receive remote monitoring services as part of the clinician’s care offering.