The Pulsenmore solution utilizes your smartphone’s processing and video streaming capabilities, communication protocols, display, and power resources. For this reason, not all smartphones meet the compatibility requirements. To insure your smartphone is compatible with the Pulsenmore Home Ultrasound check the Specification webpage.
Home » News
Resource Center
Explore our comprehensive library of clinical publications, abstracts, news, events, newsletters, marketing materials and press releases.
Discover how Pulsenmore is shaping the future of healthcare delivery
Filter By:

- Publications and review articles
Integrating routine home-ultrasound telemedicine visits into prenatal care can significantly reduce maternal anxiety during pregnancy and contribute to greater maternal attachment in individuals with a history of recurrent pregnancy loss.
- Abstracts
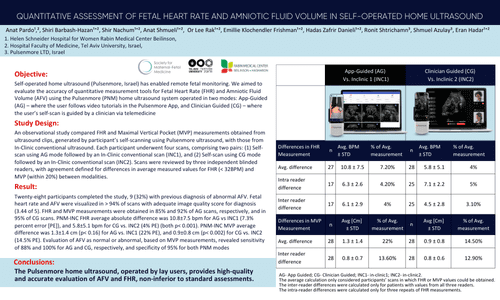
The Pulsenmore home ultrasound, operated by lay users, provides high-quality and accurate evaluation of AFV and FHR, non-inferior to standard assessments.
- Events
The 2nd Summit of the Israeli Society for HealthTech ‘will take place on September 9th, 2024 at VALLEY Kfar Saba Events that have recently shaken our world exposed the need
- News, Newsletters
We’re excited to share the latest from Pulsenmore! Discover upcoming opportunities to connect in person, learn about our recent collaborations, and explore our newest training initiatives. Don’t miss out on
- Abstracts
The INSTINCT ultrasound system is a viable solution for remote fetal assessment and could potentially lead to a cost and time alleviation. There is need for integration of Doppler-mode to
- Events
The Global Growth Engine for the Women’s Health Industry WHIS are on a mission to revolutionize the women’s health industry by uniting stakeholders from across the industry to drive innovation

the news pulse
We invite you to keep up to date with the latest updates by registering to our quarterly newsletter




FAQ
Frequently asked question
How do I know if my smartphone is supported?
Why is the device single-paitent use?
Our commitment to patient safety and reliable scans guides the device’s design and functionality. The Pulsenmore device, connecting to a patient’s personal smartphone, optimally utilizes smartphone capabilities, minimizing material and resource consumption for enhanced environmental sustainability.
1. The Pulsenmore self-scan device is designed for use within a specific timeframe and usage limit. Extending its lifetime may compromise reading integrity due to material limitations.
2. Responsibility for device maintenance and home storage after the initial period cannot be assumed, impacting transducer quality and ultrasound scan reliability.
3. The device design and validated cleaning protocol do not allow transfer between patients, to avoid cross contamination and infection.
Can the product be used in a pregnancy with multiple fetuses?
No, the product is subject to use in single pregnancies only.
Can the device increase anxiety or create dependency?
The home ultrasound device is designed to provide continuity during pregnancy care and is intended to give you peace of mind.
Can I scan without medical interpretation?
Every scan requires medical interpretation, and it’s important to wait until the files are fully uploaded. You are not expected to understand or interpret the ultrasound scans; just follow the app’s instructions to scan, and receive a response from skilled ultrasound professionals .
Can I view the scans I make through the Pulsenmore App?
You can view the scan in real-time while following the scanning instructions in the app. After the scan, the images are sent to the doctor and cannot be reviewed again.
What is the size of the generated clips?
In the App-Guided mode, the size of the zip file is about 20MB.The Clinician-Guided recorded scan is usually smaller, depending on the amount and length of clips.
How long can scans be viewed on the interface?
Data is available for review for 30 days. During this time, the HCP can download and save the scans onto the patient file. More information is available in our Terms of Use.
Can anyone access the dashboard?
No, the Clinician-Dashboard interface is an online web application that utilizes an active directory access technology (permission management tool).
How does the patient receive her Pulsenmore Scan Key Code?
The Pulsenmore Scan Key Code is generated by the HCP or admin within the clinician dashboard interface.It has unique identifiers based on the patient de-identified ID, selected mode (Clinician-Guided or App-Guided), and prescription details (frequency and time of use).The key is automatically transformed into a QR code and shared with the patient via email or SMS.
Is the device tranferable between patients?
The device is designed for a single patient use, to prevent cross contamination.
Does Pulsenmore use the data for its own purposes?
Pulsenmore utilizes service logs and technical information to improve services and enhance user experience.Pseudonymized clinical data (also referred as de-identified data in the US) is used for clinical education,training, research and development purposes only.